
This document focuses on the practical operations of noncontact temperature measurement devices and IR thermometry, and answers important questions that may arise. If you plan to use a noncontact temperature measurement device and require further advice, send us your application and we will give you the best solution.
The advantages of using IR thermometers
Temperature is the most frequently measured physical quantity, second only to time.
Temperature plays an important role as an indicator of the condition of a product or piece of machinery, both in manufacturing and in quality control. Accurate temperature monitoring improves product quality and increases productivity.
Infrared technology is not a new phenomenon it has been utilised successfully in industrial and research settings for decades but new innovations have reduced costs, increased reliability, and resulted in noncontact infrared sensors offering smaller units of measurement.
Advantages offered by noncontact temperature measurement:
1. It is fast (in the ms range). Time is saved, allowing for more measurements and accumulation of data.
2. It facilitates measurement of moving targets (conveyor processes).
3. Measurements can be taken of hazardous or physically inaccessible objects.
4. Measurements of high temperatures (up to 3000°C) present no problems. In similar cases, contact thermometers cannot be used, or have a limited life.
5. There is no interference and no energy is lost from the target. For example, in the case of a poor heat conductor such as plastic or wood, measurements are extremely accurate with no distortion of measured values, as compared to measurements with contact thermometers.
6. There is no risk of contamination and no mechanical effect on the surface of the object; thus wear-free. Lacquered surfaces, for example, are not scratched and soft surfaces can also be measured.
What to keep in mind when using an IR thermometer
1. The target must be optically (infrared-optically) visible to the IR thermometer. High levels of dust or smoke make measurement less accurate. Concrete obstacles, such as a closed metallic reaction vessel, allow for only surface measurement - the inside of the container cannot be measured.
2. The optics of the sensor must be protected from dust and condensing liquids.
3. Normally, only surface temperatures can be measured, with the differing emissivities of different material surfaces taken into account.
The infrared measuring system
An IR thermometer can be compared to the human eye. The lens of the eye represents the optics through which the radiation (flow of photons) from the object reaches the photosensitive layer (retina) via the atmosphere. This is converted into a signal that is sent to the brain. Figure 1 shows an infrared measuring system process flow.
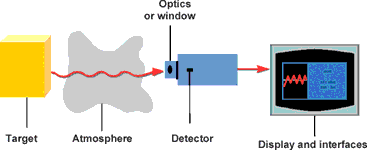
The target
Every form of matter with a temperature (T) above absolute zero emits infrared radiation according to its temperature. This is called characteristic radiation. The cause of this is the internal mechanical movement of molecules. The intensity of this movement depends on the temperature of the object. Since the molecule movement represents charge displacement, electromagnetic radiation (photon particles) is emitted. These photons move at the speed of light and behave according to the known optical principles. They can be deflected, focused with a lens, or reflected from reflective surfaces. The spectrum of this radiation ranges from 0,7 to 1000 μm wavelength. For this reason, this radiation cannot normally be seen with the naked eye. This area lies within the red area of visible light and has therefore been called 'infra'-red after the Latin. (See Figure 2)
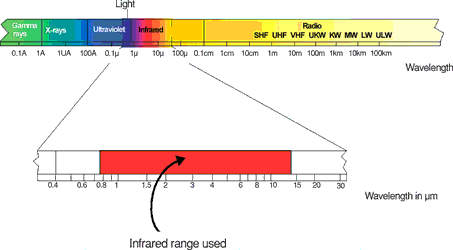
Figure 3 shows the typical radiation of a body at different temperatures. As indicated, bodies at high temperatures still emit a small amount of visible radiation. This is why everyone can see objects at very high temperatures (above 600°C) glowing somewhere from red to white.
Experienced steelworkers can even estimate temperature quite accurately from the colour. The classic disappearing filament pyrometer was used in the steel and iron industries from 1930 on. The invisible part of the spectrum, however, contains up to 100 000 times more energy. Infrared measuring technology builds on this. It can likewise be seen in Figure 3 that the radiation maximum move toward ever-shorter wavelengths as the target temperature rises, and that the curves of a body do not overlap at different temperatures. The radiant energy in the entire wavelength range (area beneath each curve) increases to the power of four of the temperature. These relationships were recognised by Stefan and Boltzmann in 1879 and illustrate that an unambiguous temperature can be measured from the radiation signal.
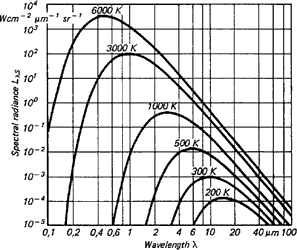
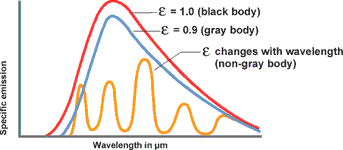
Looking at Figure 3, then, the goal should be to set up the IR thermometer for the widest range possible in order to gain the most energy (corresponding to the area below a curve) or signal from the target. There are, however, some instances in which this is not always advantageous. For instance, in Figure 3, the intensity of radiation increases at 2 μm much more when the temperature increases than at 10 μm. The greater the radiance difference per temperature difference, the more accurately the IR thermometer works. In accordance with the displacement of the radiation maximum to smaller wavelengths with increasing temperature (Wien's Displacement Law), the wavelength range behaves in accordance with the measuring temperature range of the pyrometer. At low temperatures, an IR thermometer working at 2 μm would stop at temperatures below 600°C, seeing little to nothing since there is too little radiation energy. A further reason for having devices for different wavelength ranges is the emissivity pattern of some materials known as non-grey bodies (glass, metals, and plastic films). Figure 3 shows the ideal, the so-called 'blackbody'. The relation between the real emissive power and that of a blackbody is known as emissivity ε (epsilon) and can be a maximum of 1 (body corresponds to the ideal blackbody) and a minimum of 0. Bodies with emissivity less than 1 are called grey bodies. Bodies where emissivity is also dependent on temperature and wavelength are called non-grey bodies.
Furthermore, the sum of emission is composed of absorption (A), reflection (R) and transmission (T) and is equal to one. (See Equation 1 and Figure 4).
A + R + T = 1 .............................. (1)
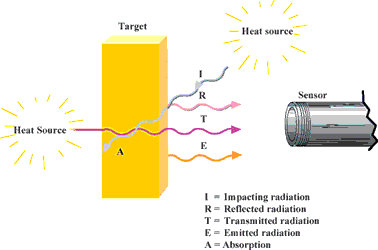
Solid bodies have no transmission in the infrared range (T = 0). In accordance with Kirchhof's Law, it is assumed that all the radiation absorbed by a body, and which has led to an increase in temperature, is then also emitted by this body. The result, then, for absorption and emission is:
A <> E = 1 - R
The ideal blackbody also has no reflectance (R = 0), so that E = 1.
Many non-metallic materials such as wood, plastic, rubber, organic materials, rock, or concrete have surfaces that reflect very little, and therefore have high emissivities between 0,8 and 0,95. By contrast, metals, especially those with polished or shiny surfaces have emissivities at around 0,1. IR thermometers compensate for this by offering variable options for setting the emissivity factor. (See also Figure 5.)
Measuring metals
The emissivity of a metal depends on wavelength and temperature. Since metals often reflect, they tend to have a low emissivity which can produce differing and unreliable results.
In such a case it is important to select an instrument which measures the infrared radiation at a particular wavelength and within a particular temperature range at which the metals have the highest possible emissivity. With many metals, the measurement error becomes greater with the wavelength, meaning that the shortest wavelength possible for the measurement should be used. (See Figure 6.)
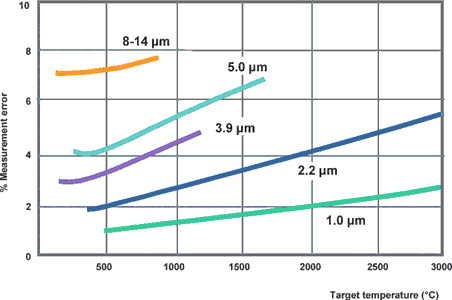
The optimal wavelength for high temperatures in the case of metals is, at around 0,8 to 1,0 μm, at the limit to the visible range. Wavelengths of 1,6, 2,2, and 3,9 μm are also possible. Good results can be achieved using ratio pyrometers in cases (eg, heating processes) where measurement is to take place across a relatively wide temperature range and the emissivity changes with the temperature.
Measuring plastics
The transmittance of a plastic varies with the wavelength and is proportional to its thickness. Thin materials are more transmissive than thick plastics. In order to achieve optimal temperature measurement it is important to select a wavelength at which transmittance is nearly zero. Some plastics (polyethylene, polypropylene, nylon, and polystyrol) are not transmissive at 3,43 μm; others (polyester, polyurethane, Teflon FEP, and polyamide) at 7,9 μm. With thicker (>0,4 mm), strongly-coloured films, you should choose a wavelength between 8 and 14 μm. If you are still uncertain, send a sample of the plastic to the manufacturer of the infrared device to determine the optimal spectral bandwidth for measurement. Almost all plastic films have reflectance between 5 and 10%.
Measuring glass
When measuring the temperature of glass with an infrared thermometer, both reflectance and transmittance must be considered. By carefully selecting the wavelength, it is possible to measure temperature of both the surface and at a depth. When taking measurements below the surface, a sensor for 1,0, 2,2, or 3,9 μm wavelength should be used. We recommend you use a sensor for 5 μm for surface temperatures. At low temperatures, 8,14 μm should be used with the emissivity set to 0,85, to compensate for reflectance. Since glass is a poor conductor of heat, and can change surface temperature rapidly, a measuring device with a short response time is recommended.
Ambient conditions
Another reason for setting up an IR thermometer for a particular spectral range only (spectral radiation pyrometer), is the transmission behaviour of the transmission path, usually the ambient air. Certain components of the atmosphere, such as vapour and carbon dioxide, absorb infrared radiation at particular wavelengths which result in transmission loss.
If absorption media is not taken into account, it can lead to a temperature displayed below that of the actual target temperature. Fortunately, there are 'windows' in the infrared spectrum which do not contain these absorption bands. Typical measuring windows are 1,7 μm, 2,5 μm, 3,5 μm and 8,14 μm. Since the manufacturers have already furnished infrared measuring devices with atmospheric correction filters, the user is spared such worries.
Thermal radiation in the environment surrounding the target should likewise be taken into account. The higher temperatures of the furnace walls could lead to errors in temperature measurement on metal pieces in an industrial furnace. The possible effect of the ambient temperature has been taken into consideration by many infrared measuring devices, with compensation built in. The other possibility is a too-high temperature being displayed for the target. A correctly set emissivity, along with automatic ambient temperature compensation from a second temperature sensor ensures extremely accurate results.
Dust, smoke, and suspended matter in the atmosphere can result in contamination of the optics and, therefore, in false measured values. In order to prevent deposition of suspended matter, optional air-blowing attachments are offered. These are usually screw-on pipe connections with a compressed air supply. The air ensures overpressure in front of the optics, thus keeping contaminating particles at bay. If a great amount of dust or smoke is created during the measurement procedure and affects the result, then ratio pyrometers should be used.
IR sensors are electronic devices and can only work within certain operating temperature ranges. Some sensors allow an upper limit of 85°C. Above the permitted operating temperature, air or water-cooling accessories must be used and there must be special connection cables for the application of high temperature. When using water-cooling it is often useful to use it in conjunction with the air-blowing attachment to prevent formation of condensation on the optics.
| Tel: | +27 11 608 1551 |
| Email: | [email protected] |
| www: | www.randci.co.za |
| Articles: | More information and articles about R&C Instrumentation |

© Technews Publishing (Pty) Ltd | All Rights Reserved