

Moisture content is very often the only parameter used to define moisture conditions in hygroscopic products. It influences many of the physical and mechanical properties of materials and even their selling price. Moisture content is, and will remain, of great interest to both manufacturers and their customers. However, moisture content does not address all problems (product stability, shelf life for example).
Water activity (aw) or equilibrium relative humidity (ERH) quantifies the active part of the moisture content or 'free water' as opposed to the total moisture content which also includes `bound water'. Water activity (aw or ERH) not only determines the stability of moisture content (and of the parameters it influences) but is also a decisive factor in various problems where moisture content is of less interest. Theoretically, moisture content can be used to determine aw and vice versa, however, in practice this possibility is very limited and both parameters should be measured separately - depending on the problem to be solved.
The importance of aw or ERH has long since been recognised by the food and paper industries. Other industries, such as the pharmaceutical and chemical industries, show an ever-increasing interest for aw measurements in addition or as an alternative to the usual moisture content measurements. This application note discusses some of the most important applications of aw in various industries and shows how it should be measured.
Moisture content and water activity (aw)
The moisture content of a product is usually defined as the percent weight of water content in relation to the dry weight of the sample. Water activity or equilibrium relative humidity measures the water-vapour pressure generated by the water present in a hygroscopic product.
Aw = p/ps and ERH = 100 X aw, where:
p = water-vapour pressure above the product surface
ps = water-vapour pressure above the surface of pure water at the product temperature
Hygroscopic products may absorb water in different ways: sorption and chemical reaction, sorption and formation of a hydrate, water binding by surface energy, diffusion of water molecules in the material structure, capillary condensation formation of a solution etc. Therefore, water molecules present in a product are not all retained by the product with the same strength. In fact, only part of the total moisture content can be exchanged between the product and its surroundings under normal conditions.
Moisture content includes both an immobilised part (eg, water of hydration) and an active part. This explains the lack of accuracy and reliability of most methods for measuring %H2O. The quantity of water found in a product sample depends on both the method of measurement and on the way it is applied. The active part of moisture content - or free water - is usually measured in terms of its vapour pressure, expressed as percent relative humidity generated in equilibrium with the product sample in a closed system at constant temperature.
Water activity (or ERH) indicates the degree of freedom of the water absorbed in a material and shows dimensions, structure, cohesion, agglomeration properties as well as electrical and chemical properties better than moisture content does. The effect of this water on physical properties, such as differences in water-vapour pressure, and not moisture content govern the interchange of water between a product and its surroundings (air, packaging, other products etc). Therefore, the stability of the moisture content of a product is determined by any difference between ERH and % rh in the surrounding air or by any difference in aw or ERH with other products.
Sorption isotherms
At equilibrium, the relationship between %H2O and aw of a hygroscopic material can be graphically represented by a curve called a sorption isotherm. For each value of aw a sorption isotherm shows the corresponding value of H2O at a given constant temperature.
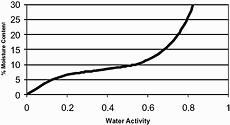
Due to the complexity of sorption phenomena, sorption isotherms cannot be calculated in advance and have to be experimentally determined for each single product. The practical usefulness of sorption isotherms is severely limited by several factors:
* Sorption isotherms are valid only for a single product.
* Sorption isotherms are affected by any variation in product.
* Composition (eg, products of natural origin).
* Sorption isotherms show a difference between absorption and desorption curves (sorption hysteresis).
* Sorption isotherms are experimentally determined, unfortunately most methods used for measuring % H2O are neither very accurate or reliable.
From the above it should be clear that moisture content very often cannot be used to accurately determine aw and vice versa. The practical usefulness of sorption isotherms is limited to products whose previous moisture history is known and which have a defined composition and a stable temperature.
The slope of most sorption isotherms is such that a small variation in %H2O corresponds to a large variation in aw. Due to this fact, moisture content cannot be used to make a rough estimate of aw. However, aw can often be used for moisture checks such as the check of a moisture content limit.
In practice, moisture content and water activity should be considered as two independent parameters.
aw (or ERH) in the paper industry
The importance of ERH to the paper industry is extensively explained in our bulletin 'Paper and Climate'. When both the ERH and temperature of paper, as well as the %rh and temperature of the surrounding air are kept within adequate limits a great number of problems can be minimised or even eliminated:
* Static electricity phenomena.
* Deformations of paper during storage.
* Poor register in colour printing.
* Dusting problems in offset machines.
* Excessive drying times of ink.
FOGRA has studied these problems and has set up general recommendations concerning ERH. It is worth noting that a given value of ERH corresponds to different values of %H2O for different paper types. No general recommendation could be given in %H2O.
aw in the food industry
The concept of 'free' as opposed to the total, including 'bound' water, has gained wide acceptance in the food processing industry. Water activity exerts a decisive influence on such phenomena as change in colour, taste and aroma, food poisoning and spoilage (shelf life), loss of vitamins etc. (Total moisture content has very little to do with this).
Water activity in foods can be controlled by using various additives (salts, sugars etc), by using satisfactory packaging materials, by maintaining favourable maturation and storage conditions. Water activity measurements are increasingly frequent in food research and development as well as in production quality control. On-line measurements are possible to a certain extent: water activity is defined at static equilibrium, a condition that is not always met in the process line itself. The great importance of water activity in foods is illustrated by a few examples in the following paragraphs.
Growth of micro-organisms
Water activity indicates the amount of water in the total water content that is available to micro-organisms. Each species of micro-organism (bacteria, yeast and mould) has its own minimum aw value below which growth is no longer possible.
This limit corresponds to the 'suction power' of the various organisms, ie, to the osmotic pressure they create and which must be higher than in the aqueous phase of food, to absorb from the food the water needed for metabolic activity and growth (see table below). By measuring the aw value of foodstuffs it is possible to determine which micro-organisms will not be able to develop on them.
The US Food and Drug Administration has adopted the concept of aw for establishing moisture limits beyond which certain types of food are considered susceptible to invasion by mould and bacteria. Under this concept, foods with diverse water binding capabilities can be considered as a group (this would not be possible with an %H2O concept). A water activity specification has already been incorporated in one FDA regulation (Fed. Reg. Vol. 44 no. 52 March 16 1979). Similar standards will very probably be introduced into Europe as well.
Typical growth limits function of aw:
Most bacteria: 0,91 to 0,95
Most yeast: 0,88
Most mildew: 0,80
Halophile bacteria: 0,75
Osmiophil yeast: 0,70
Xerophile mildew: 0,65
Chemical stability
Water activity control is an important factor for the chemical stability of foods. Most foodstuffs contain carbohydrates and proteins and are therefore subject to non-enzymatic browning reactions (Maillard reaction). The Maillard reaction gets stronger at increasing aw values and reaches its peak at aw = 0,6 to 0,7 with further increase of aw this reaction gets rapidly weaker.
The spontaneous autocatalyctic breaking of the molecular chains of fats is strongly influenced by aw. This kind of food spoilage increases at high aw values. Even at low aw values, foods with a fat content acquire a rancid taste after being stored for some time. Foods in which the action of glyceride creates fatty acids with short molecular chains are particularly affected by this kind of spoilage: they produce a strong and disagreeable smell. The conservation of foodstuffs is influenced by numerous changes occasioned by oxidation: colour change of carotin, oxidation of myoglobin in meat, oxidation of proteins and vitamins etc. The oxidation of fats and other food components decreases sharply at aw values below 0,2.
Enzymatic stability
Most enzymatic reactions are slowed down at aw values below 0,8. Some of these reactions occur even at very low aw values. However, as many foodstuffs are thermally treated during their processing, enzymatic spoilage is usually of very little importance.
Drying process
With a few exceptions, the molecular activity of water-vapour (ie, pressure) in hygroscopic materials increases with temperature at roughly the same rate as the increase in pressure of saturated water vapour. In fact water activity shows only limited temperature dependence.
Increasing the temperature of air in a dryer neither decreases its water-vapour pressure nor increases its 'hygroscopicity'. Warm air increases the water-vapour pressure generated by the product to be dried by increasing its temperature. The drying enthalpy and therefore the energy necessary to dry a product is strongly influenced by water activity. For pure water (ie, aw = 1,0) the evaporation enthalpy amounts to 2300 to 2500 kJ/kg. This explains why the last part of the moisture content is so expensive to extract when drying.
The speed at which water can be removed by evaporation from a product is directly proportional to the active surface of the product and to the difference in water-vapour pressure between the product and its surroundings. Differences in moisture content have nothing to do with this! Calculating and controlling drying process requires the knowledge of the relationship between aw and %H2O (sorption isotherms). As mentioned in paragraph 3, this relationship is especially hard to determine in the case of products of natural origin (variable product composition). In practice it is necessary to consider that sorption isotherms correspond to conditions of static equilibrium. Therefore, differences with theoretical calculations based on sorption isotherms will occur.
Optimising a drying process can only be done by experimentation. As already shown, moisture content measurement (air or product) does not give an accurate image of the drying process itself. Measurement of the temperature drop in the drying air-flow (psychometric effect) gives results that are very dependent on the conditions at the inlet. Besides this, such a measurement neither takes into account the sorption properties of the product to be dried nor the effect of temperature on the water vapour generated by the product. The dynamic moisture and temperature equilibrium that exists in a dryer cannot be considered as strictly equivalent to water activity. Nevertheless, measuring both the relative humidity and temperature of the product (eg, %rh and °C at the outlet of a fluid bed dryer) gives the closest image of the drying process. Under consideration of temperature the relative humidity established by dynamic equilibrium in the dryer can be compared to the ERH (aw) that the product should have after drying. In this way the nature of the product and in particular the effect of temperature on its water-vapour pressure can be taken into account automatically.
Drying a product to a given moisture content can be a costly exercise. From the point of view of energy conservation it is worth asking if water activity rather than moisture content should be considered in the definition of the product moisture condition after drying. Whenever product quality and stability are the main factors to determine what a dryer should do, the answer should be positive. Significant energy savings as well as better quality control will result from such a decision.
Without optimisation of the instrument design to meet product and application specific requirements, it is often impossible to achieve accurate and reliable measurements. Consequently, Rotronic offer a wide range of instruments, which, if correctly selected, will provide many years of service without the need for repeated repair or calibration. Rotronic is represented locally by Action Instruments SA.
| Tel: | +27 11 403 2247 |
| Email: | [email protected] |
| www: | www.aisa.co.za |
| Articles: | More information and articles about Action Instruments SA |

© Technews Publishing (Pty) Ltd | All Rights Reserved