
The flotation process can separate a desired mineral such as copper, nickel, molybdenum or zinc from ore. Flotation can separate the desired mineral from waste materials or from another mineral.
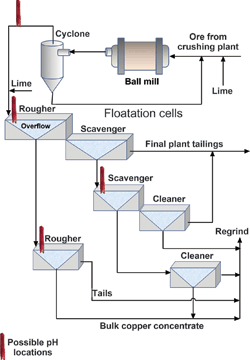
The process starts with crushed ore received by a ball mill and reduced in particle size to the consistency of sand. Next, the ground ore from the mill travels as high-solid slurry to a cyclone. The cyclone separates the fine and heavier particles, which return to the ball mill for further grinding. The cyclone overflow continues on to a series of flotation cells that separate the desired mineral from other materials.
Air injected into the flotation cells, along with foaming agents, creates a bubbly froth. An added chemical called a collector acts on the lighter mineral, causing it to attach to bubbles and rise to or near the surface. The float material then overflows or is skimmed off, while the bottom material goes on to another separation process or to waste. The ability of a mineral to float depends on certain conditions. One important flotation variable is pH, which affects:
1. The form of many ionic species present.
2. The charge state of the mineral surfaces.
3. The form of many collectors.
Copper processing example

A high pH is important in the processing of copper ore to maximise this mineral‘s flotation properties. Most copper ore slurries tend to be acidic. Plants typically add lime to the slurry in the mill or flotation circuit, increasing its alkalinity. A typical copper operation contains many flotation cells, often with a pH sensor in each cell. The number of sensors and their locations vary from plant to plant, being largely dependent on solution chemistry. Sensor life and maintenance quickly becomes important economic factors.
The pH sensor in any mineral processing slurry should be mounted to provide sufficient velocity past the sensor to minimise lime scale and material build-up. However, the location must trade off flow velocity with the possibility of abrasion to the sensor glass and body. Additionally, the pH sensor should be located at a point that is representative of cell solution.
Some plants monitor pH further upstream of flotation in the mill circuit. A typical mill circuit installation point would be in the cyclone overflow. Plants more generally mount the pH sensor in the flotation basin or the overflow line. Mounting in the overflow has the advantage of not totally submersing the back of the sensor, provided overflow depth remains constant. Care should be taken that the sensor is mounted deep enough to contact liquid rather than the top froth, which has a high percentage of air. Most sensors have a submersible design with a rigid mounting.
The ABB solution: TB556 for pH measurement
The fluid in a flotation circuit or flotation feed will always have a high percentage of solids. The pH sensor must be rugged to stand up to the inevitable abrasion by slurry particles and build-up of scaling resulting from lime additions. In the case of scaling or particle accumulation, hydraulic cleaners have been used successfully. The periodic jet of fluid prevents a build-up of pulp or scale.
To resist abrasion of the pH electrode and plugging of the reference junction, flotation plants commonly select the ABB TB556 for this application. This sensor has an integral, patented Next Step Solid State reference. The sensor’s large annular liquid reference junction provides added surface area that’s less susceptible to plugging. The solid state reference design minimises reference contamination, resulting in a more stable and accurate measurement. And a flush flat-glass pH electrode surface reduces the adherence of particles, abrasion, and breakage.
For more information contact Jacolize Goosen, ABB South Africa, +27 (0)10 202 5000, [email protected], www.abb.com/za
| Tel: | +27 10 202 5000 |
| Email: | [email protected] |
| www: | www.abb.com/za |
| Articles: | More information and articles about ABB South Africa |

© Technews Publishing (Pty) Ltd | All Rights Reserved